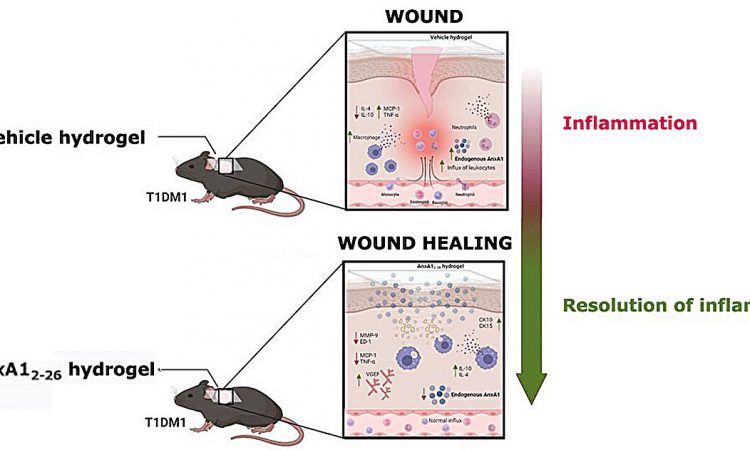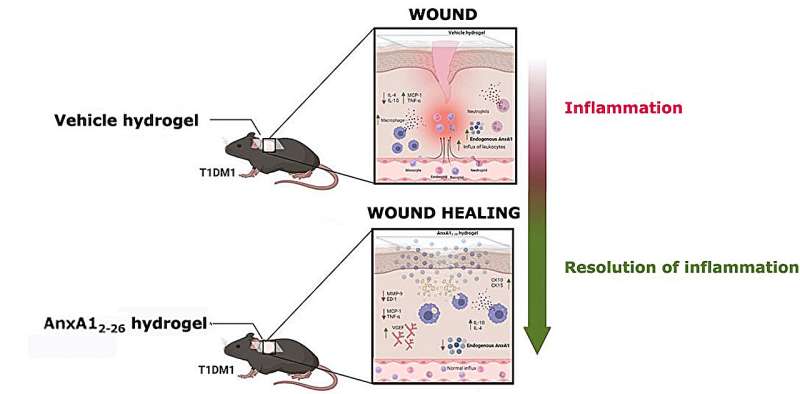
Researchers in São Paulo state, Brazil, have developed a low-cost anti-inflammatory hydrogel that in future could help treat chronic skin lesions such as those often seen in people with diabetes. They report the results of animal tests in an article published in the journal Biomedicine & Pharmacotherapy.
According to the International Diabetes Federation, Brazil ranks sixth among countries with the most cases of diabetes, which has reached epidemic proportions and become the fifth most frequent cause of death in the world. Some 17.7 million Brazilians suffer daily from the metabolic alterations caused by impaired insulin secretion and action, such as nephropathy (kidney damage), neuropathy and poor wound healing—one in five diabetics are estimated to develop chronic wounds such as foot ulcers.
In healthy people, injury of the skin immediately triggers a sequence of events that lead to healing. Bleeding is controlled by platelet aggregation, providing binding sites for coagulation and cell proliferation. This is followed by the formation of new blood cells and collagen deposition. In diabetics, however, high blood sugar levels (hyperglycemia) increase production of reactive oxygen species, which exacerbate inflammation and impair blood vessel formation.
Biological hydrogels have successfully been used to accelerate wound healing, providing a moist and sterile environment. In this study, researchers at São Paulo State University (UNESP) and the Federal University of São Paulo (UNIFESP) developed a formula based on annexin A1 (AnxA1), a protein involved in regulating inflammation and cell proliferation, as evidenced by the group’s previous research. The findings of this study show that the hydrogel can modulate the wound microenvironment and favors tissue regeneration.
The effects of the hydrogel containing AnxA12–26 (a peptide synthesized using amino acids 2 through 26) were observed in mice with induced type 1 diabetes and skin lesions made with a biopsy punch. The number of inflammatory cells was seen to decrease three days after the wounds were made. After 14 days, the wounds healed completely. In the control group, which comprised diabetic mice treated with hydrogel that did not contain the peptide, acute wound inflammation continued beyond the third day.
Immunohistochemical analysis showed an improvement in tissue regeneration due to proliferation of keratinocytes (cells that play a key role in restoring skin homeostasis and re-epithelization), reduced amounts of macrophages (immune cells that dampen inflammation, clear cell debris and coordinate tissue repair) and increased levels of vascular endothelial growth factor (VEGF), boosting the formation of new blood cells.
The cytotoxicity assay showed excellent biocompatibility and confirmed that the hydrogel is safe for use in treating diabetic wounds. “Our hydrogel is highly absorbent, providing the right amount of moisture to promote healing, which actually takes place faster than without it. It’s an effective option to expand the therapeutic arsenal for treating diabetic wounds,” said Sonia Maria Oliani, last author of the article.
Oliani is a professor at UNESP’s Institute of Biosciences, Letters and Exact Sciences (IBILCE) and UNIFESP’s Program of Graduate Studies in Functional and Structural Biology.
Other advantages of the hydrogel developed by the researchers in São Paulo are easy production and low cost, a key factor in South and Central America, where the annual cost of treating diabetes exceeds USD 65.3 billion.
The product has the potential to be used in other applications. The researchers are currently testing its use in treating oral mucosa wounds.
More information:
Monielle Sant´Ana et al, Annexin A12–26 hydrogel improves healing properties in an experimental skin lesion after induction of type 1 diabetes, Biomedicine & Pharmacotherapy (2023). DOI: 10.1016/j.biopha.2023.115230
Journal information:
Biomedicine & Pharmacotherapy
Source: Read Full Article